Reserch in Pentaferte
Research follows the constantly evolving market needs and demands for innovation from end users. The world of medical devices has the critical aspect of innovation and quality and, on the basis of these pillars, the objective of providing users with the solution to their problems, in a safe, reliable way takes shape.
Investing in research means improving our medical culture and offering it to those who, by using Pentaferte products, can increase the level of wellbeing and safety of patients and ill people.
The production process
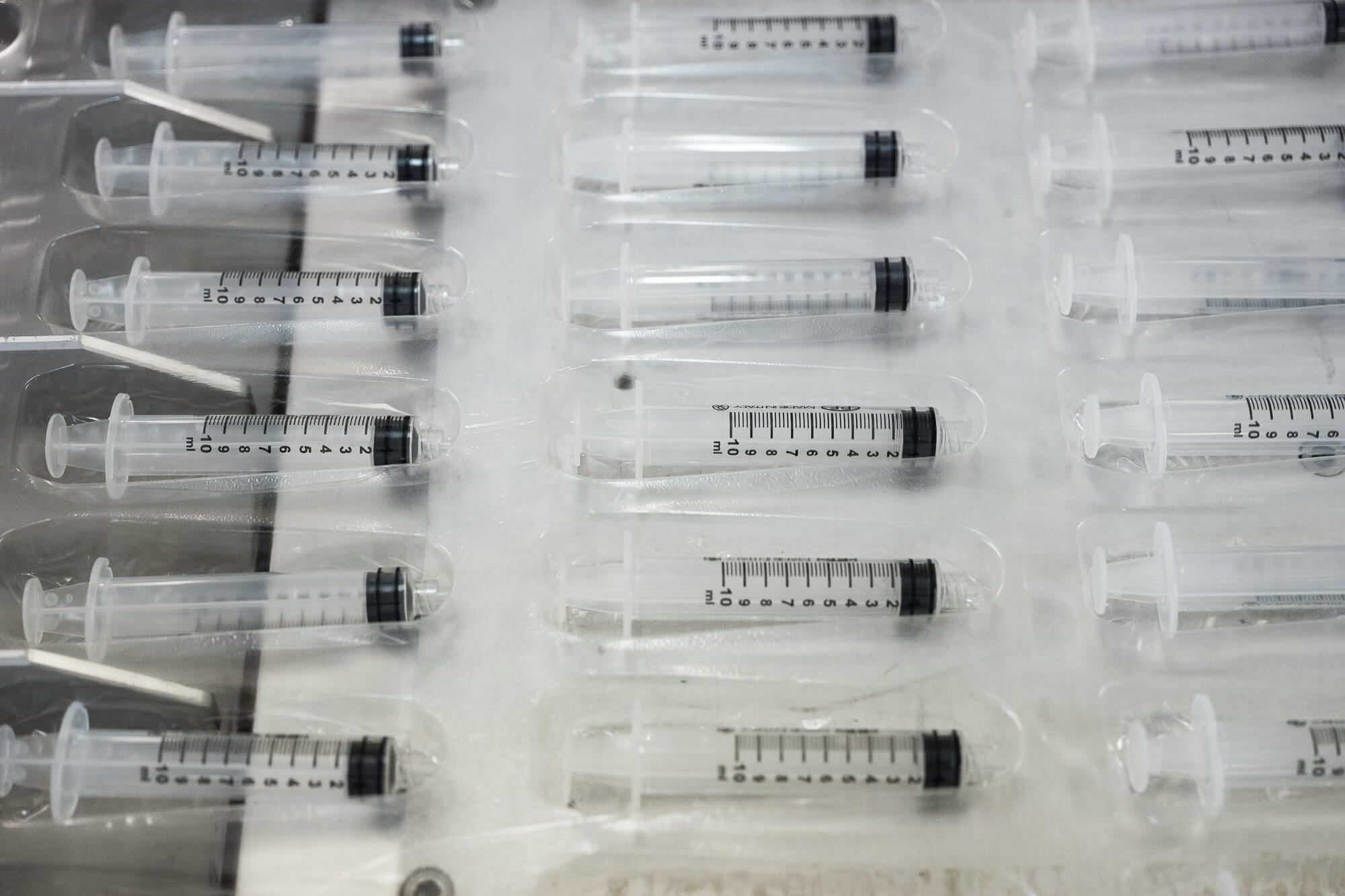
The first stage of production is the moulding that leads to the production of rods, cylinders, barrels and needle covers from pure propylene or a mixture of polypropylene and dye, previously mixed.
Once past the control stage, the semi-finished products are brought into the clean room where the following take place:
- the automatic graduation of the neutral cylinders, which produces screen-printed, silicone-coated cylinders;
- the automatic syringe assembly stage, which leads to the production of the syringes starting from the needles, rods, graduated cylinders and plungers;
- the automatic packaging that creates syringes packaged in a heat-sealed blister pack made with film on medical grade paper.
At this point in the production process, the devices leave the clean room because the product, no longer in direct contact with persons and the environment, has passed the critical stage of possible contamination and can move on to the manual packaging stage: here the semi-finished product is placed inside cartons and boxes, in turn placed into larger boxes.
Then comes the sterilisation stage with ethylene oxide, which ensures the full and total sterility of each device. This is followed by the degassing stage, which enables a reduction in residual ETO present in the product at levels permitted by applicable legislation.
Only then does the production process come to an end and the devices are brought into the finished products warehouse.